Specific Heat Capacity
_____________________________________________________________________________________
What is Specific Heat Capacity?
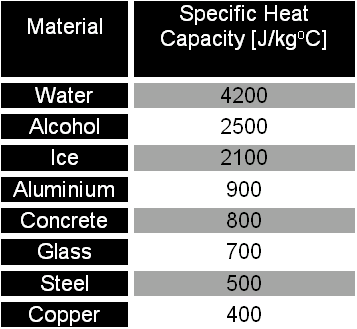
Specific Heat Capacity is the amount of energy (in Joules) needed to increase the temperature of 1 kg of a substance by 1 degree Celsius. The higher the value the more energy that needs to put in. Using this information, it is possible to calculate how much energy needs to be out into a substance to increase it temperature by a specific amount.
The equation for this is: energy transferred = mass x specific heat capacity x temperature change
This question will most likely to appear in the exam, so make sure you learn this equation!
The symbols for the above equation are as follows:
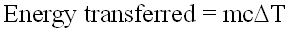
Specific Heat Capacity Chart
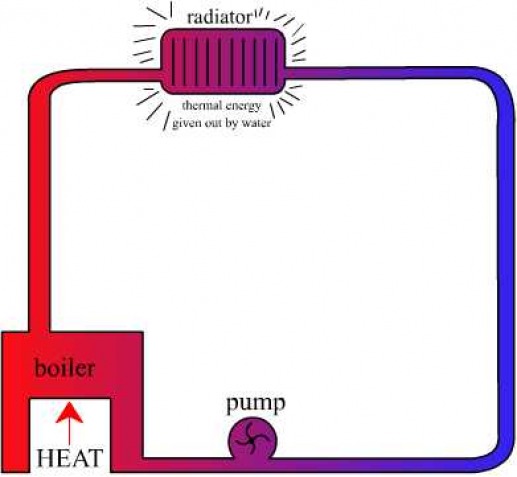
Applications
Because water has a high specific heat capacity, it is a useful substance for storage and carrying thermal energy, such as in central heating systems. It is also used in cooling systems in cars; the water removes unwanted thermal energy to the radiator from the engine.
Latent Heat
Latent heat is the amount of energy that has to be transferred to 1 kg of a substance to change its state. Latent Heat is the amount of energy that is realised or absorbed by a substance in the form of heat when it is changing state. The two main types of latent heat are latent heat of fusion (melting substance) and latent heat of vaporisation (boiling substance). An example of latent heat fusion is an ice cube at room temperature. When the ice cube is melting, it is absorbing energy in the form of heat but its temperature does not change, the energy absorbed is called the latent heat of fusion.
The equation for working out the amount of energy released from a substance with a known mass goes as follows:
Energy transferred = mass x specific latent heat
Energy transferred = mL
Latent Heat of Fusion of Ice: 330,000J/Kg
Latent Heat of Vaporisation of Water: 2,300,000J/Kg